- Home
- About AIRA
- Workgroups
- Ongoing Efforts
- Events
- Repository
- Members Only
ValidationOverview Following the stages of Testing & Discovery and immunization information system (IIS) Assessment, Validation is a summary stage to formally acknowledge and recognize IIS and IIS vendor products that are progressing toward or achieving alignment with community-selected measures and tests. As of spring 2024, IIS and IIS vendor products can achieve Validation in five content areas: Transport, Submission (VXU)/Acknowledgment (ACK), Query (QBP)/Response (RSP), Clinical Decision Support (CDS), and Data Quality Incoming/Ongoing (DQI). Why Participate?
IIS and IIS vendors can update their participation settings for Validation at any time in the Aggregate Analysis Reporting Tool (AART) Testing and Recognition
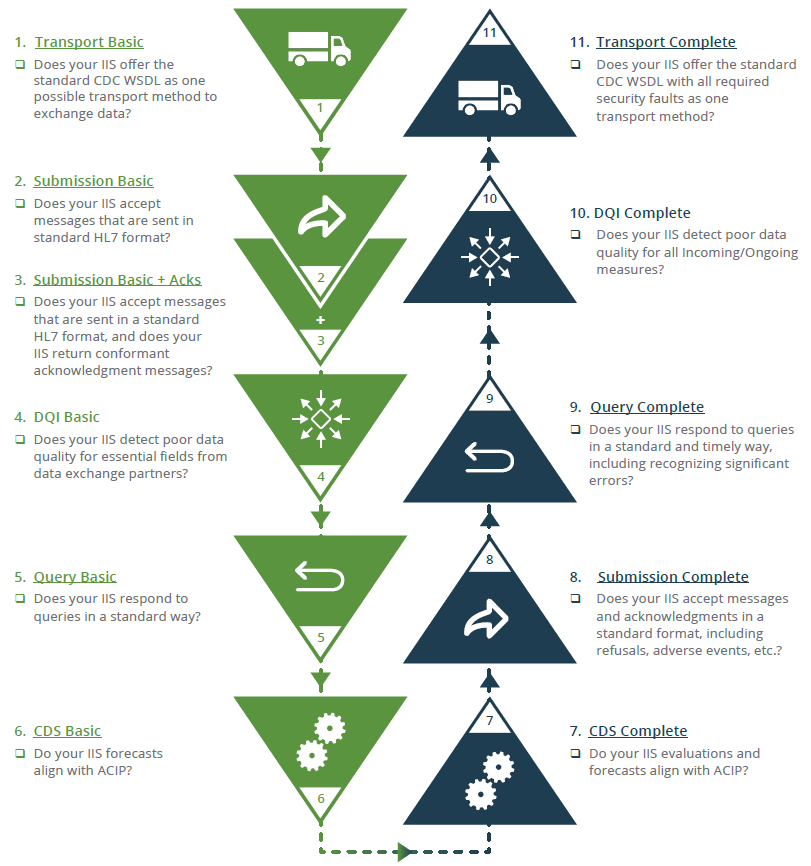
Measurement & Improvement Validation Priority OrderNot sure where to start? The graphic below suggests a prioritized path for IIS to better align with the M&I Initiative supported by the Centers for Disease Control and Prevention (CDC) and AIRA. The full two-page document can be found in the AIRA repository. The links within the document for each step seen in the image below will bring up Validation Reports that display IIS results. None of these steps should represent stopping points in the process. If your IIS program cannot make progress on one step, simply move on to the next. Log in to AART to find more detailed information specific to your IIS.
Validation by Content AreaThis content area examines the level of alignment with the formally defined transport specification, SOAP/Web Services and the CDC Web Services Definition Language (WSDL). When any two systems connect to exchange data, they must use an agreed upon transport layer to connect. Otherwise, communication and the exchange of data will be impossible. An expert panel made up of subject matter experts from across the IIS and stakeholder community selected the SOAP/Web Services and CDC WSDL as the IIS Standard in 2012.
Submission (VXU) and Acknowledgment (ACK) This content area focuses on Submission and Acknowledgment messages to an IIS. VXU, or vaccine update, is an HL7 message that contains demographic and vaccination data that is submitted to an IIS by an outside EHR. ACK, or acknowledgment, is an HL7 message used to relay information back to the EHR regarding the status of the VXU. SUB/ACK Resources:
Query (QBP) and Response (RSP) This content area focuses on Query by Parameter (QBP) HL7 message submissions that are sent to an IIS and Response (RSP) HL7 message submissions that are returned by an IIS in response to a querying system. The QBP is used to query data from the IIS and contains basic demographic data about a patient. The sending system wants to know if the IIS has any demographic and/or vaccination data on said patient, so it “asks” the IIS to respond to the QBP message. Once the IIS receives a QBP, the IIS attempts to match the demographic information contained in the QBP message with data in the IIS. It then sends back an appropriate response (RSP) HL7 message telling the sending system if the patient was found in the IIS and what information (if any) was found. QBP/RSP Resources:
Clinical Decision Support (CDS) This content area focuses on Clinical Decision Support (CDS) tools designed to automatically determine the recommended immunizations needed when a patient presents for vaccination. These recommendations are developed by the Advisory Committee on Immunization Practices (ACIP). The measures for this content area focus on three CDS concepts that can be returned via HL7 messages: Evaluation Status, Earliest Date, and Recommended Date. Each IIS is assessed on capability to return a concept and accuracy if the concept is returned based on age of the patient. CDS Resources:
Data Quality Incoming/Ongoing (DQI) This content area focuses on the IIS ability to detect data quality issues on a per-message basis. The IIS will be presented with messages that contain intentional data quality errors (e.g., vaccination date before date of birth) and the IIS is expected to detect these errors. The IIS will also be assessed on the ability to retain (and return) vaccination events from submission to query without changing the meaning (e.g., NDC mapped to CVX, return key information about a vaccination event). DQI Resources:
Additional Resources
Updated: 3/29/24 |